Research
Mechanisms of innate immune evasion by vaccinia virus and how this impacts on the immunogenicity and efficacy of this virus as a vaccine vector
 Research in the lab of Prof. Geoffrey Smith focuses on many aspects of host-pathogen interactions using the poxvirus vaccinia virus (VACV) as a model. My particular research focuses on the evasion of innate immunity by VACV. To establish a productive infection viruses must counteract the immune system, in particular the early host innate response and VACV encodes an arsenal of proteins that inhibit innate immune signalling (in particular the activation of key transcription factors such as NF-κB and IRF3). By inhibiting these pathways the virus limits the production of the anti-viral interferons as well as pro-inflammatory cytokine/chemokines. By studying the mechanisms of VACV inhibition of host signalling our aim is to enhance current understanding of the innate immune response, how it is activated and regulated, with the potential identification of novel drug targets for diseases caused by excessive inflammation. What is particularly interesting is that despite encoding multiple inhibitors of both NF-kB and IRF3 activation, which act at various stages of these innate signalling pathways, these viral proteins are not redundant as removal of a single gene attenuates the virus in vivo in mouse models of VACV infection. This is important as poxviruses (e.g. modified virus Ankara (MVA)) are being intensely studied as vaccine vectors to protect against a number of infectious agents including HIV and malaria, particularly as they are good stimulators of innate and adaptive immunity, including both cellular and humoral responses. A major goal of this research is to make these vectors as safe as possible, whilst still retaining their high immunogenicity profile. Recent research in our lab, as well as other groups, has demonstrated that removal of certain immune inhibitory genes (particularly innate immune inhibitors) not only attenuates VACV, making it safer, but also enhances immunogenicity. I am particularly interested in understanding how and why removal of innate immune modulators affects the ensuing adaptive and memory immune responses, positively impacting the immunogenicity profile of these potential vaccine vectors.
Research in the lab of Prof. Geoffrey Smith focuses on many aspects of host-pathogen interactions using the poxvirus vaccinia virus (VACV) as a model. My particular research focuses on the evasion of innate immunity by VACV. To establish a productive infection viruses must counteract the immune system, in particular the early host innate response and VACV encodes an arsenal of proteins that inhibit innate immune signalling (in particular the activation of key transcription factors such as NF-κB and IRF3). By inhibiting these pathways the virus limits the production of the anti-viral interferons as well as pro-inflammatory cytokine/chemokines. By studying the mechanisms of VACV inhibition of host signalling our aim is to enhance current understanding of the innate immune response, how it is activated and regulated, with the potential identification of novel drug targets for diseases caused by excessive inflammation. What is particularly interesting is that despite encoding multiple inhibitors of both NF-kB and IRF3 activation, which act at various stages of these innate signalling pathways, these viral proteins are not redundant as removal of a single gene attenuates the virus in vivo in mouse models of VACV infection. This is important as poxviruses (e.g. modified virus Ankara (MVA)) are being intensely studied as vaccine vectors to protect against a number of infectious agents including HIV and malaria, particularly as they are good stimulators of innate and adaptive immunity, including both cellular and humoral responses. A major goal of this research is to make these vectors as safe as possible, whilst still retaining their high immunogenicity profile. Recent research in our lab, as well as other groups, has demonstrated that removal of certain immune inhibitory genes (particularly innate immune inhibitors) not only attenuates VACV, making it safer, but also enhances immunogenicity. I am particularly interested in understanding how and why removal of innate immune modulators affects the ensuing adaptive and memory immune responses, positively impacting the immunogenicity profile of these potential vaccine vectors.
Publications
Smith GL, Benfield CT, Maluquer de Motes C, Mazzon M, Ember SW, Ferguson BJ, Sumner RP (2013) Vaccinia virus immune evasion: mechanisms, virulence and immunogenicity. Journal of General Virology.
Ferguson BJ, Benfield CTO, Ren H, Lee V, Frazer GL, Strnadova P, Sumner RP, Smith GL (2013) Vaccinia virus protein N2 is a nuclear IRF3 inhibitor that promotes virulence. Journal of General Virology 94, 2070-2081.
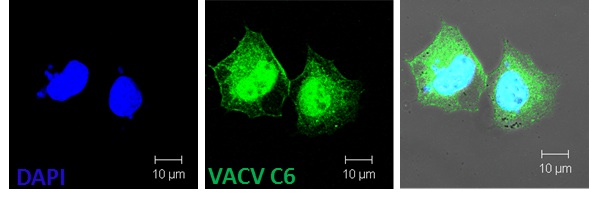
Sumner RP, Ren H, Smith GL (2013) Deletion of immunomodulator C6 from vaccinia virus strain Western Reserve enhances virus immunogenicity and vaccine efficacy. Journal of General Virology 94, 1121-1126.
Mansur DS, Maluquer de Motes C, Unterholzner L, Sumner RP, Ferguson BJ, Ren H, Strnadova P, Bowie AG, Smith GL (2013) Poxvirus targeting of E3 ligase b-TrCP by molecular mimicry: A mechanism to inhibit NF-kB activation and promote immune evasion and virulence. PLoS Pathogens 9(2): e1003183.
Unterholzner L, Sumner RP, Baran M, Ren H, Mansur DS, Bourke NM, Randow F, Smith GL, Bowie AG (2011) Vaccinia Virus Protein C6 is a Virulence Factor that Binds TBK-1 Adaptor Proteins and Inhibits Activation of IRF3 and IRF7. PLoS Pathogens 7:e1002247.
Sumner, R. P., Ren, H. & Smith, G. L. Deletion of immunomodulator C6 from vaccinia virus strain Western Reserve enhances virus immunogenicity and vaccine efficacy. J Gen Virol.



